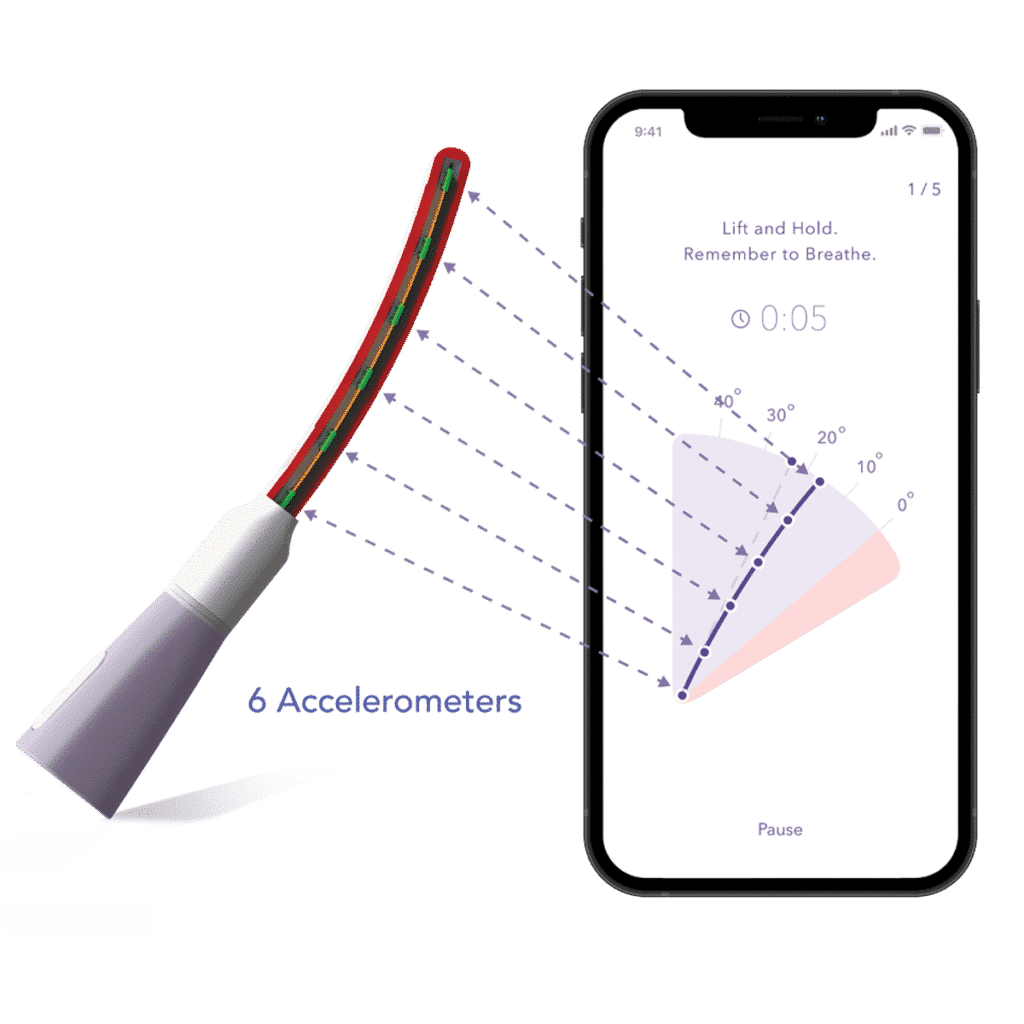
How Leva Works
The Leva Pelvic Health System combines motion-based, multi-sensor technology embedded in a flexible intra-vaginal probe with integrated software to provide Supervised Pelvic Floor Muscle Therapy (PFMT) for the treatment of female pelvic floor disorders.
UI Indication
The Leva Pelvic Health System is an FDA-cleared, prescription-only medical device indicated for the treatment of stress, mixed and mild to moderate urgency urinary incontinence (including overactive bladder) in women. The Leva Pelvic Health System is a Class II medical device and was authorized by the FDA through a 510(k) Pre-Market filing (K213913). The Leva Pelvic Health System also received Breakthrough Device Designation (Q211109) from the FDA on October 28, 2021, for the chronic fecal incontinence indication.
Pharmacy Code
60002099440
HCPCS CODE
S9002
Clinical Outcomes
Pivotal Data
Published in Obstetrics & Gynecology (“The Green Journal”), our pivotal 8-week randomized controlled trial (RCT) with 299 women demonstrates that pelvic floor muscle training guided by Leva delivers better UI symptom improvement than an at home program alone.2 Primary outcomes were change in stress urinary incontinence (SUI) episodes on a 3-day bladder diary and change in Urogenital Distress Inventory (UDI-6) score.
- The 3-day bladder diary results showed that the median number of SUI episodes at 8 weeks was significantly fewer in the Leva arm than in the Control arm. Most Leva users saw an 80% reduction in leakage episodes.2
- For the (UDI-6) scores on symptom relief, the Leva users reached a significantly greater score improvement compared to the Control arm.
- The Leva users saw statistically significant symptom improvement as early as 4 weeks into treatment.2
Read the full study, published in ACOG’s Green Journal in April 2022, here

Long Term Data
Published in the International Urogynecology Journal, an 18- & 24-month long term follow-up to the 8-week pivotal, followed the same 299 participants and UI symptom improvement persisted for two years, despite Leva users no longer using Leva. Findings show durable results lasting for two years. Leva is a scalable option for practices to implement for first line UI care.
Read the full study, published in the International Urogynecology Journal in January 2024, here
Real World User Evidence
Our peer-reviewed real-world evidence study of 947 Leva users, published in JMIR Publications in June 2024, found:
- Leva is effective for stress, mixed and urgency urinary incontinence (including overactive bladder).
- 74% of Leva users see significant UI symptom relief.
Read the full study, published in JMIR Publications in June 2024, here
Budget Impact Model Study
A peer-reviewed study in the Journal of Medical Economics shows that using the FDA-cleared Leva Pelvic Health System as a first-line treatment for female urinary incontinence can significantly reduce health plan costs. Key results from the analysis include:
- In a United States commercial health plan with 1 million members, 334,191 were adult women, of whom 31,438 (9.4%) were treated for UI over 24 months. The study compared health-plan costs for two groups: a group receiving current clinical practice for first-line UI care (CCP) and a group in which 15% of patients received Leva, while all others received CCP.5
- The total estimated 24-month cost per treated patient was $10,447 for the group in which 15% received Leva versus $11,267 for the 85% receiving CCP. The total cost for the Leva group ($10,447) includes the intervention cost of $2,100.
- This resulted in total projected savings for the 1-million-member health plan of > $25.7 million over 24 months, or $1.07 PMPM.
Read the full study, published in the Journal of Medical Economics in May 2025 here
Leva Pelvic Health System Overview and Clinical Evidence Summary
Discover how the FDA-cleared Leva Pelvic Health System is transforming urinary incontinence therapy. Explore our clinical evidence, market adoption, and provider utilization in this comprehensive overview.
Plan Savings With Leva
Publications
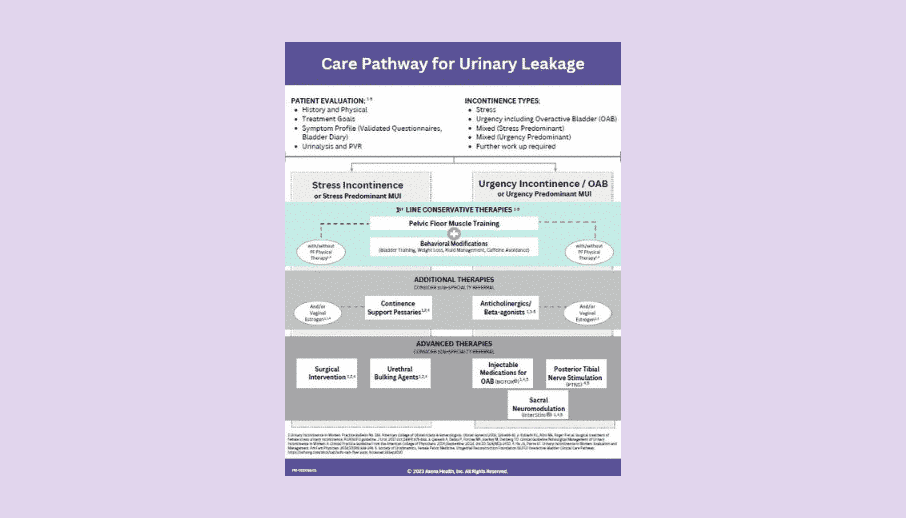
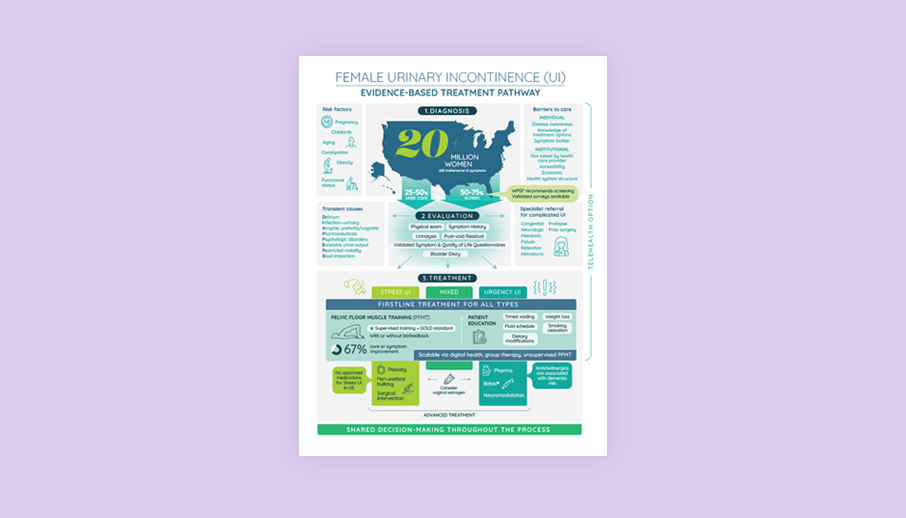
Several publications support the finding that UI is costly and undertreated, and there is a real lack of documented first line care.